Over 100 million Americans have chronic pain.
That’s almost one of every three people in the U.S. As individuals live longer and treatments for cancer are more successful, the number of individuals living with chronic pain will continue to increase.
Scientists working in many fields, including neurophysiology and genetics, seek to discover how acute pain persists to become chronic pain. In the meantime, healthcare clinicians are challenged to treat the pain experienced by individuals with persistent pain.
A month ago you started treating your patient with stage II non-Hodgkin’s lymphoma with radiation and chemotherapy. Prior to treatment, your patient reported a 3 month history of jaw clicking without and with pain, and frequent headaches.
During this visit, you will explore how your patient’s pain experiences evolve, how this relates to what we know about the transition of acute to chronic pain, and the promising research being done to discover how acute pain persists to become chronic pain.
This visit also gives you the opportunity to check your knowledge of the mechanisms of pain processing, how these relate to the mechanisms of action for analgesics and non-pharmacologic (biobehavioral) treatments, and apply this knowledge to reduce and/or relieve your patients evolving pain experiences.
Consider again the Biopsychosocial Model of Pain.
Think about what factors influence the transition from acute to persistent and chronic pain.
The OPPERA Study
The Orofacial Pain Prospective Evaluation and Risk Assessment study (OPPERA) is the first large prospective research specifically designed to examine and identify biopsychosocial, environmental and genetic factors that contribute to the onset and persistence of Temporomandibular Disorders (TMD).
Funded by the National Institutes of Dental and Craniofacial Research, a seven year prospective cohort study was initiated by a group of internationally recognized scientists with expertise in epidemiology, pain mechanisms, TMD diagnostics, functional genomics, statistical genetics, bioinformatics, and biostatistics. Approximately 85% of the cost of treating TMD is associated with treating patients who develop persistent pain and dysfunction.
The primary goal of the OPPERA study is to identify the psychological and physiological risk factors, clinical characteristics and associated genetic mechanisms that influence the development of a persistence of TMD (i.e., factors that lead to the onset of TMD). This study also seeks to characterize the biological pathways through which these genetic variations causally influence TMD risk.
The methodological goals were to:
- Undertake a prospective cohort study of over 3, 000 initially TMD-free individuals recruited from major ethnic and racial groups at 4 study sites, identifying 204 incident cases of first-onset-TMD during a 5-year period.
- Undertake a case-control study by recruiting about 200 individuals with chronic symptomatic TMD identified during cohort recruitment.
- Measure in both groups potential predictors of TMD risk, including both non-causal predictors (Aim 1) and etiological factors (Aims 2–4), analyzing their individual and joint effects using a conceptual, causal model for TMD development based on previously published research.
The epidemiological data will be used to address four specific aims to determine if:
- Socio-demographic characteristics thought to be predictive of TMD (e.g., younger age, female sex, white race) are associated with elevated risk of first-onset TMD and increased odds of chronic TMD.
- Heightened responsiveness to noxious stimuli, delivered using standardized pain measurement procedures, together with biological processes associated with this heightened responsiveness (e.g. resting arterial blood pressure) contribute to the risk of first-onset TMD.
- Pre-existing psychosocial profiles commonly observed with TMD and other chronic pain conditions (e.g., depression, anxiety, perceived life stress and stressful life events) are associated with elevated risk of first-onset TMD.
- Genetic variations in candidate genes that influence pain amplification and psychological profiles are associated with elevated risk of first-onset and chronic TMD; and if so, to identify the biological, psychological and clinical pathways through which these genetic variations causally influence TMD risk.
Temporomandibular disorders (TMD) are common. Five percent of US adults report TMD-type pain (6% of women, and 3% of men), while an examination-survey of a representative sample of females in New York City found that 10% had examiner-diagnosed TMD. This 2 to 1 ratio of female to male disorder prevalence in the general population and 8 to 1 in clinical settings suggests identifiable risk factors.
Numerous risk factors have been, implicated from cross-sectional studies; including: joint and muscle trauma, anatomical relationships (e.g. skeletal and occlusal), pathophysiological factors (e.g., bone and connective tissue disorders, hormonal differences, sensitization of peripheral and central nervous system pain processing pathways) and psychosocial factors (e.g., depression, anxiety, and perceptual responses to stressors) However, it is unclear if these potential risk factors represent causal influences for developing TMD.
Based on the potential risk factors identified to date and the biopsychosocial model, Dr. Samuel Dworkin and colleagues created a model of causal influences contributing to onset and persistence of TMD.
This model proposes that TMD is influenced by two sets of intermediate phenotypes: psychological distress and pain amplification. Interactions between the two phenotypes and the environment further contribute to onset and persistence of painful TMD. Genetic regulation of biological mechanisms that determine expression of phenotypes is modeled as a distal but significant contributor. Time is not depicted in the model because time is a requirement for the development of chronic pain, and its effects occur implicitly on a third dimension.
This model may be translatable to other pains that persist from acute onset to chronic pain diseases.
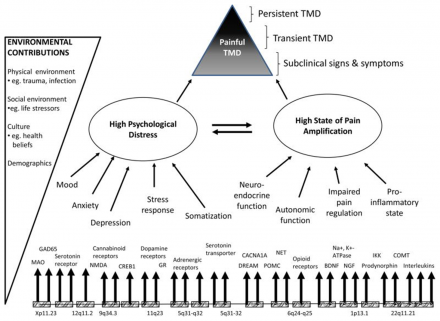
*Image source: Orofacial Pain Prospective Evaluation and Risk Assessment Study - The OPPERA Study (PDF Download Available). Available from: https://www.researchgate.net/publication/221717188_Orofacial_Pain_Prosp… [accessed Sep 16, 2016]
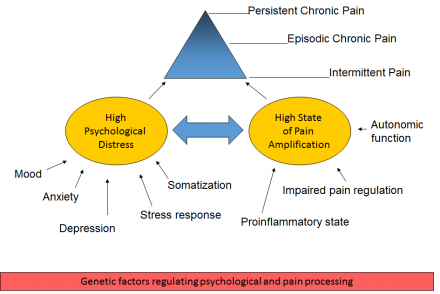
This model further defines the biological, psychological, and social influences on pain.
So now, consider again the Biopsychosocial Model of Pain. What factors influence the transition from acute to persistent and chronic pain in your patient’s case?
Treatment Check - 1 Month into Treatment
Chronic pain is often defined by time, including as little as one month of persistent pain.
Is your patients pain acute, recurrent acute, persistent or chronic pain?
One month into treatment with chemotherapy:
Jaw pain significantly better, but fatigue and headaches continue.
(Jaw Pain: This is persistent/chronic or recurrent acute since its lasted more than one month, but is not always present.)
(Headaches: This is persistent/chronic or recurrent acute since its lasted more than one month, but is not always present.)
What additional pharmacologic treatment would you recommend at this time?
Hover over your choices to compare the rationale with the subject matter experts.
Acetaminophen 500 mg every 4 hours PRN headaches
(Acetaminophen is a first line treatment for headaches. Monitor amount and frequency of use to avoid toxicity. Also, monitor for fevers, since this patient may be immunocompromised from chemotherapy. Frequent acetaminophen administration may mask a fever, the only sign of possible infection in patients with neutropenia from chemotherapy.)
Ibuprofen 400 mg PO every 6 hours PRN headaches
(Ibuprofen is an NSAID which would be another first line treatment for headaches, but may not be the best choice for this patient, who is actively getting chemotherapy.
Chemotherapy introduces the possibility of increased risk of bleeding due to thrombocytopenia (chemo side effect), and NSAIDs inhibit platelet aggregation.)
Gabapentin 200 mg PO TID.
(Gabapentin is not indicated for headaches and is only used for migraine prophylaxis when other medications with stronger evidence of effectiveness have failed to prevent frequent migraines.)
Treatment Check - 2 Months into Treatment
Two months into treatment with chemotherapy, daily radiation begins:
Jaw pain continues but is better than before diagnosis. (This is persistent/chronic or recurrent acute, since its lasted more than one month but is not always present.)
Oropharyngeal mucositis pain
(This is acute at this time due to concurrent radiation and chemotherapy. If mucositis is the result of chemotherapy alone, the mucositis improves as the white blood cell count recovers.)
Throat pain is >10 of 10 unbearable, unable to eat or swallow own saliva due to pain.
Tongue pain intensity is 8 of 10.
General pain – difficult to describe but she emphasizes it is real and nothing seems to help.
(If patient reports this pain has persisted for less than a month, it is acute pain.)
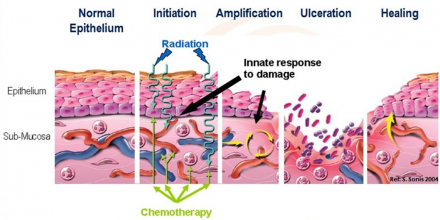
*Image source: "Management of Oral Mucositis in Patients with Cancer" (2008).
Articles - Research. Paper 181. http://digitalcommons.uconn.edu/uchcres_articles/181
Recall how the treatments below address mechanisms of pain.
Reports ice helps jaw pain – somewhat (The mechanisms beyond pain relief following brief applications of cold have not been elucidated.
With more prolonged application, cold results in slowing of nerve conduction velocity, particularly in unmyelinated (C) and thinly myelinated (A-delta) primary afferent nerves. Pain signaling carried along these afferents is slowed, limiting transmission to the dorsal horn, or spinal trigeminal nucleus for orofacial pain, and diminishing pain sensation.)
Acetaminophen 500 mg every 4 hours PRN for headache - minimally reduces headache severity (inhibits prostaglandin synthesis in the central nervous system.)
The following provide minimal relief, some with side effects:
- Magic mouthwash (lidocaine, maalox, & diphenhydramine); burns for 20 minutes
- Artificial saliva (Caphosol); mix 1 blue and 1 clear vial, swish and spit 4 to 10 times/day
- Lansoprazole (Prevacid); 30 mg PO daily
- Other medications converted to IV form in the hospital for severe throat pain:
- Ondansetron (Zofran) 8 mg IV every 8 hrs PRN; for nausea
- Lorazepam (Ativan) 0.5 mg IV every 6 hrs PRN; for nausea or anxiety
Test Your Knowledge
Question 1
Of the following analgesic medications, which is considered the drug of choice for the treatment of prolonged moderate to severe pain for the patient with cancer?
Question 2
The usual time to peak effect for traditional analgesics (acetaminophen, NSAIDs, and opioids) given orally is:
The decision is made to add an opioid for pain control and discontinue acetaminophen.
Hydromorphone (Dilaudid) IV PCA 0.2mg/demand as often as every 6 minutes, with a maximum of 1mg/hour.
The patient reports tongue and throat pain decreased to 4 of 10 within 24 hours after hydromorphone IV PCA started.
Question 3
After the initial recommended dose of opioid analgesic, subsequent doses should be adjusted in accordance with the individual patient's response.
Then, 24 hours later, mucositis is improving and patient is able to change to oral hydromorphone, 4 mg. PO every 3 hours PRN pain.
Question 4
The recommended route of administration of opioid analgesics to the patient with prolonged cancer-related pain is:
Treatment Check - 6 months into treatment
Post-radiation therapy, your patient has lost 10 kg due to:
- mucositis and throat pain (Recurrent acute due to chemotherapy at this time.)
- jaw/facial pain (Persistent/chronic since its lasted more than one month and now seems to always be present)
- nausea, vomiting, and anorexia
Your patient reports bilateral foot pain
(Persistent/chronic; anticipate this pain will last longer than one month and chemotherapy may need to be altered) toes to mid-feet, and dysesthesias of the feet, specifically numbness and tingling, “like pins and needles when your feet fall asleep.” Pain wakes your patient from sleep and there is nothing she can do to relieve it.
Your patient demonstrates a steppage gait due to foot drop, as well as loss of sensation to vibration, allodynia, and hyperesthesia to pin prick. These new findings are believed to be due to vincristine chemotherapy.
Would you anticipate your patient’s pains will remain acute, recurrent acute, persistent or chronic pain?
Treatments continued:
- Hydromorphone - 4 mg PO every 3 hours PRN for jaw and mucositis pain
- Lansoprazole (Prevacid) - 30 mg PO daily
- Ondansetron (Zofran) - 8 mg IV every 8 hours PRN for nausea
- Lorazepam (Ativan) - 0.5 mg IV every 6 hrs PRN for nausea or anxiety
- Sennoside - 2 tabs 1-2 times daily to achieve at least 1 BM/day
- Polyethylene glycol - (Miralax) 17 grams 1-2 times daily to achieve at least 1 BM/day
Your patient reports taking 3 to 4 doses of hydromorphone a day, and this medication helps the jaw and mucositis pain, but not the foot pain.
Test Your Knowledge
Question 1
What additional pharmacologic treatment would you recommend to treat the new onset neuropathic bilateral foot pain at this time? Hover over your choices to compare the rationale with the subject matter experts.
Question 2
Non-drug interventions (e.g. guided imagery, biofeedback, transcutaneous electrical nerve stimulation (TENS) etc.) are very effective for mild-moderate pain control but are rarely helpful for more severe pain.
Question 3
Combining analgesics and non-drug therapies that work by different mechanisms (e.g. using acetaminophen, anticonvulsant, topical anesthetics, TENS unit, etc.) may result in better pain control with fewer side effects than using a single analgesic agent.
Treatment Reminders for Pain that Persists
Your patient explains that her pain is better controlled but she wakes up in the middle of the night every night due to pain. This pain is then relieved in 30 to 60 minutes by hydromorphone.
If the patient is experiencing around the clock pain that is relieved by an opioid, consider scheduled treatment or use of extended release or controlled release opioid for pain control.
Your patient continues to have bothersome allodynia and dysesthesias.
The dosages of anticonvulsants and antidepressants used to treat neuropathic pain must be optimized to balance efficacy and adverse effects. Titration upwards should be anticipated at the beginning of treatment; and titration downwards (weaning) is necessary when the medication is no longer needed. When opioids are deemed no longer needed, weaning is also necessary to avoid opioid withdrawal when patients have been consistently receiving opioids for more than 5 to 7 consecutive days.
Patient completes chemotherapy for cancer treatment.
Whenever possible consider the potential risk-benefit of adding acetaminophen or NSAID for analgesia and opioid-sparing effect.
Key Points as Pain Persists
Genetic predisposition to decreased or increased sensitivity to pain (genetic variants) results in physiologic differences including alterations in how pain is transmitted from the periphery and/or modulation of pain signals in the brain and descending pain modulatory system.
Learned behavioral responses to pain, such as catastrophizing and somatization, can cause increased distress and heightened stress response. Other psychological factors may contribute to the risk of chronic pain, such as increased perceived stress, anxiety and depression, by enhancing pain signaling throughout the central nervous system.
With ongoing pain, the peripheral and central nervous system becomes progressively responsive to sensory input resulting in hypersensitivity.
Glial activation and decreased function of interneurons, which modulate pain signals, play an important role in central sensitization; a state in which peripheral nociceptive input is no longer necessary to activate central pain mechanisms.
Increased afferent input and enhanced neuronal excitability lead to neuroplasticity that changes the patterns of activation within higher-order pain processing regions of the brain. Changes in neuroplasticity reflect the development of a “pain memory.”
The perception of pain is maintained through central pain mechanisms without any required input from the periphery. These changes can be seen in functional Magnetic Resonance Imaging (MRI) in patients before they experience pain as compared to once they develop chronic pain, as well as between patients who have or do not have chronic pain.
Test Your Knowledge
Question 1
Non-drug interventions (e.g. guided imagery, biofeedback, transcutaneous electrical nerve stimulation (TENS), etc.) are very effective for mild-moderate pain control but are rarely helpful for more severe pain.
Question 2
Parents should NOT be present during painful procedures.
Question 3
Analgesia for chronic cancer pain should be given:
Question 4
Which of the following drugs are useful for treatment of cancer pain?
Question 5
The usual duration of analgesia of Morphine IV is 4-6 hours.
Question 6
The most likely explanation for why a child/adolescent with pain would request increased doses of pain medication is:

