Pain
Definition
“An unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described in terms of such damage.” (IASP)
Reference
International Association for the Study of Pain (IASP) Definitions and Terminology
Significance of Pain
- Pain is adaptive
- Alerts us to danger
- Motivates escape and avoidance learning
- Motivates recuperation
- Pain is subjective
- Influenced by expectations and emotions
Take a Minute
- Why is it important to know that pain is subjective?
- How can pain be influenced by expectations and emotions?
Assessing Pain: Pain Scales
- Scale of 1-10
- Verbal Descriptor Scale
- Wong-Baker Facial Grimace Scale
- Activity Tolerance Scale
Universal Pain Assessment Tool
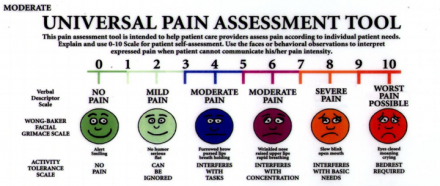
For more, go to http://ebnurse.org/pain-management/
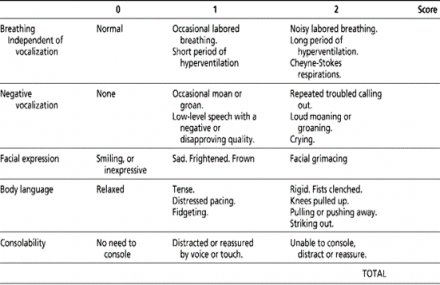
Patient Case
VPK is a 90-year-old male with a diagnosis of late-stage Alzheimer’s Disease. The patient has difficulty describing his pain to the healthcare team. You can use the following to determine the level of pain in this patient:
- Rate of breathing
- Vocalization
- Facial expression
- Body language
Pain Processing
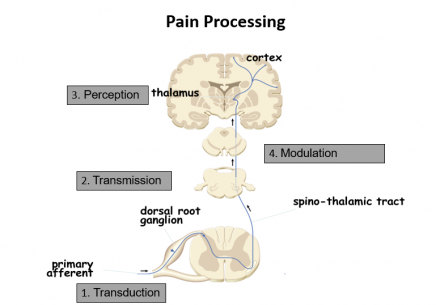
Modified from Cleveland Clinic. Cancer pain.
4 Steps of Pain Processing
- Transduction
- Information from the periphery
- Nociception
- Transmission
- Spinal cord to the brain
- Perception
- How the brain processes the information
- Modulation
- How is the pain signal “handled” as it descends from the brain to the spinal cord
Transduction
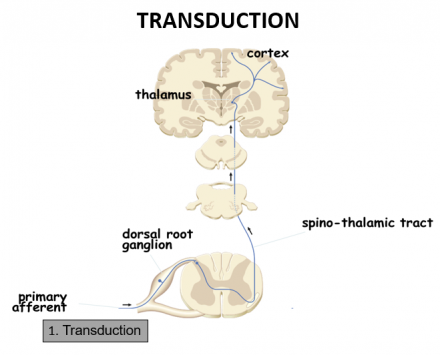
- Nociceptors (free nerve endings) of Aδ and C fibers are exposed to noxious stimuli
- Noxious stimuli include:
- mechanical (pressure, swelling, incision)
- thermal (burn, scald)
- chemical (neurotransmitters, toxic substance, ischemia, infection)
- Chemical messengers released by damaged cells may be targets for pharmacotherapy
- bradykinin
- serotonin
- substance P
- prostaglandins
Transmission
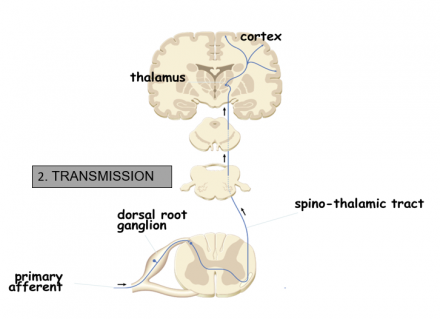
- Begins at the site of transduction traveling along the nociceptor fibers to the dorsal horn in the spinal cord
- Aδ and C fibers terminate in the dorsal horn
- Excitatory neurotransmitters are released propagating the signal from the spinal cord to the brain
- The pain impulse is transmitted from the spinal cord to the brain stem and thalamus via the ascending pathways
- The brain does not have a discrete pain center, so the impulse is processed at the thalamus and then directed to additional brain regions
Peripheral Pain Fibers: Transmission
- Primary afferents
- A-δ fibers (fast pathway)
- Thick, myelinated, fast conducting neurons
- Mediate the feeling of initial fast, sharp, highly localized pain
- Activated by mechanical or thermal pain
- C fibers (slow pathway)
- Very thin, unmyelinated, slow-conducting
- Mediate slow, dull, more diffuse, often burning pain
- Polymodal (mechanical, thermal, chemical)
- Projections to brain centers responsible for emotional responses: displeasure, anxiety, etc.
Afferent Sensory Fibers: A-δ and C
| Feature | A-δ fibers | C fibers |
|---|---|---|
| Structure | Myelinated | Unmyelinated |
| Amount | 10% | 90% |
| Source | Thermal and mechanical stimuli | Polymodal: mechanical, thermal and chemical stimuli |
| Speed | Fast traveling, 5-10 m/sec | Slow traveling 0.6-2 m/sec |
| Sensory quality | Sharp, stinging , cutting, pinching | Dull, burning, aching |
Adapted from: Copstead –Kirkhorn L-E, Banasik J. Pathophysiology 4th ed. Elsevier Publishing, 2009
Ascending Pain Pathway
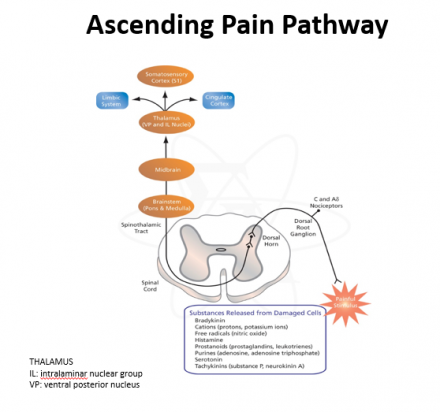
Ascending Pain Review
- Nociceptors respond to intense thermal, mechanical, and chemical stimuli
- Located in all pain sensitive regions of the body
- Activated by chemicals released during tissue damage and inflammation, causing transmission of action potentials
- Axons of these neurons carry the signal into the cord, release neurotransmitters that activate pain transmission neurons in the dorsal horn of the spinal cord
- Pain transmission neurons carry this signal to various regions of the brain where it is processed and evaluated
Perception
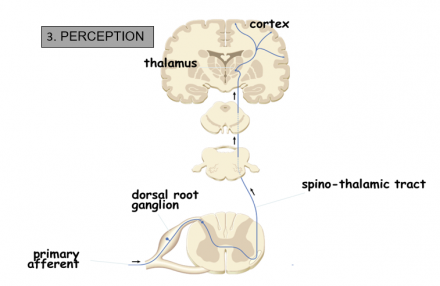
Image modified from Cleveland Clinic.
- Pain becomes a conscious multidimensional experience
- Activation of multiple cortical regions
- The reticular system:
- responsible for the autonomic and motor response to pain (i.e. quickly removing a hand when it touches a hot stove)
- Somatosensory cortex:
- important for the interpretation of pain information being processed
- identifies the intensity, type and location of the pain sensation and relates the sensation to past memories and experiences
- Limbic system:
- responsible for the emotional and behavioral responses to pain
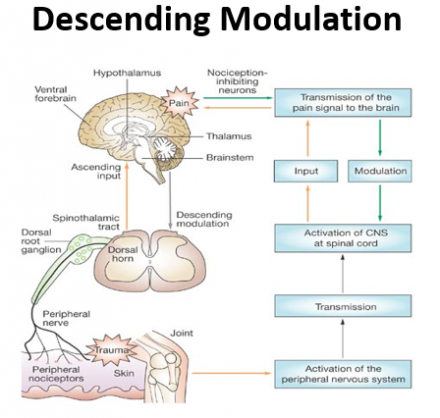
Modulation
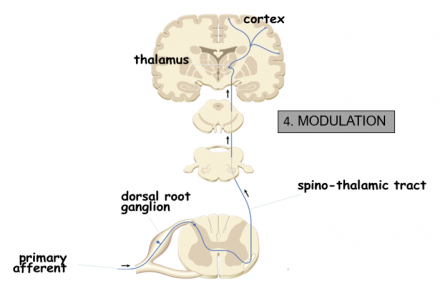
Image modified from Cleveland Clinic.
- Beginning at the brain, the descending pain pathway utilizes inhibitory signals to block the pain signal from being processed in the spinal cord
- Descending inhibition involves the release of inhibitory neurotransmitters that produce analgesia
- Inhibitory neurotransmitters involved with the modulation of pain include:
- Endogenous opioids, serotonin, norepinephirine , GABA
- Analgesics commonly used in the treatment of pain may produce their pharmacological actions through these neurotransmitter pathways
Descending Modulation
Additional resources on nociception:
Dublin AE, Patapoutian A. Nociceptors: The sensors of the pain pathway. J Clin Invest
Bingham B, Ajit SK, Blake DR, Samad TA. Nature Clin Practice Rheumatology 2009; 5:28-37.
Neurotransmitters: Modulation
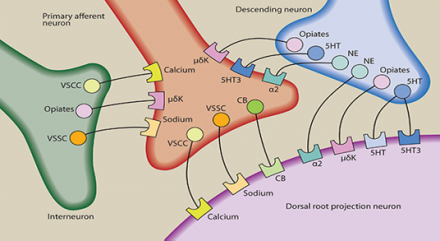
Adapted from Stahl's essential psychopharmacology. 2008
There are many neurotransmitters and their corresponding receptors in the dorsal horn. Neurotransmitters in the dorsal horn may be released by primary afferent neurons, by descending regulatory neurons, by dorsal horn projection neurons, and by interneurons. Neurotransmitters present in the dorsal horn that have been best studied in terms of pain transmission include substance P (NK receptors-not shown), endorphins (μ opioid receptors), norepinephrine NE (alpha 2 adenoreceptors) and serotonin (5HT, 5HT3 receptors). Several other neurotransmitters are also believed to be involved in pain and neuronal health such as calcium (VSCC-voltage sensitive calcium channel) sodium (VSSC-voltage sensitive sodium channel) and cannabinoids (CB).
For more: Collaborative Medication Management – Rational Prescribing. Normal physiology: The sensation of Pain.
Types of Pain
- Nociceptive
- Neuropathic
- Inflammatory
For more: University of Wisconsin School of Medicine and Public Health. Pain Management.
Nociceptive Pain
Pain that arises from actual or threatened damage to non-neural tissue and is due to the activation of nociceptors
Can be divided in to:
- Somatic
- Visceral
Somatic
Pain arising from tissues such as skin, muscle, tendon, joint and bone
well localized.
Visceral
Pain arising from the internal organs. Often diffuse or referred.
Neuropathic Pain
- Pain caused by a lesion or disease of the somatosensory nervous system
- Neuropathic pain is a clinical description and not a diagnosis
- Patients may describe the pain as burning, electric, searing, tingling, and migrating or traveling
- Some causes of neuropathic pain include:
- amputation (phantom limb pain), herpes zoster, AIDS (peripheral neuropathy), diabetic neuropathy, fibromyalgia, and cancers that affect the spinal cord, among others
For more: International Association for the Study of Pain (IASP)
Terms Associated With Neuropathic Pain
- Hyperalgesia
- increased pain from a stimulus that normally provokes pain
- Allodynia
- pain due to a stimulus that does not normally provoke pain
- Sensitization
- increased responsiveness of nociceptive neurons to their normal input, and/or recruitment of a response to normally sub-threshold inputs
- mediated by chemical messengers
For more: International Association for the Study of Pain (IASP)
Inflammatory Pain
- A localized protective reaction of tissue to irritation, injury, or infection, characterized by pain, redness, swelling, and sometimes loss of function
- Activates the nociceptive pathway
- Examples of inflammatory pain include:
- appendicitis, rheumatoid arthritis and inflammatory bowel disease
Time Course of Pain
- Acute
- typically does not last longer than three to six months, and resolves when the underlying cause of pain has been treated or has healed
- Chronic
- persists despite the fact that the injury has healed
- pain signals persist beyond three to six months and can last for years
Treatment Options
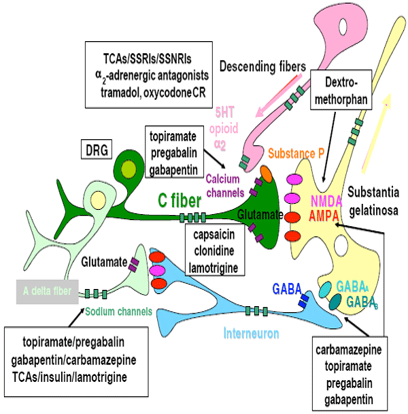
- Right analgesic(s) - based on type and severity of pain and co-morbidities.
- Right route - when possible the oral route is preferred
- Right dose - understand there is great inter-patient variability
- Right dosing interval - scheduled dosing vs PRN dosing
- Right message - patient expectations, use of nondrug interventions
For more: University of Wisconsin School of Medicine and Public Health.
Pain Management: Treatment Planning.
Opioid Agents: Use, Abuse, and Management
Pain Management
Morphine
- Is a potent agonist at the mu opioid receptor
- Multiple routes of administration
- Metabolized by hepatic cytochrome P450 enzymes and glucuronidation into a number of active metabolites
- Excreted by renal elimination
- May cause respiratory depression and death
- May lead to tolerance and dependence
- May cause constipation and urinary retention
- Discontinuation may lead to withdrawal symptoms
- DEA Schedule II drug
Oxycodone
- Is an agonist at the mu opioid receptor
- Good oral bioavailability
- Metabolized by hepatic cytochrome P450 enzymes into a number of active metabolites
- Some of its metabolites, such as oxymorphone, are more potent than oxycodone
- Excreted by renal elimination
- May cause respiratory depression and death
- May lead to tolerance and dependence
- May cause constipation and urinary retention
- Discontinuation may lead to withdrawal symptoms
- DEA Schedule II drug
Hydrocodone
- Is an agonist at the mu opioid receptor
- Good oral bioavailability
- Metabolized by hepatic cytochrome P450 enzymes into a number of active metabolites
- Some of its metabolites, such as hydromorphone, are more potent than hydrocodone
- Excreted by renal elimination
- May cause respiratory depression and death
- May lead to tolerance and dependence
- May cause constipation and urinary retention
- Discontinuation may lead to withdrawal symptoms
- Often used in combination with acetaminophen, ibuprofen, or aspirin
- DEA Schedule II drug
Withdrawal
Clonidine
- Used to treat opioid withdrawal and other indications (e.g., high blood pressure)
- Acts by decreasing sympathetic nervous system responses, reduces hypertension and tachycardia associated with opioid withdrawal
- Is an agonist at the α2A-adrenergic receptor, which is found predominantly in the brain
- Good oral bioavailability
- Some of the orally-administered clonidine is metabolized by the liver and some is excreted unmetabolized by the kidneys
- Sudden discontinuation can result in hypertension
- Clonidine should not be used in pregnant women
Antispasmodics
- Antispasmodics are medications that suppress muscle spasms
- Dicyclomine, also known as dicycloverine, and hyoscyamine are antispasmodics that act as antagonists at the acetylcholine receptors
- Side effects, due to the anticholinergic properties of these medications, include dry mouth, nausea, blurred vision, etc.
Loperamide
- Used to treat diarrhea
- Does not cross the blood-brain barrier
- Is an agonist at the mu opioid receptor
- Metabolized by hepatic cytochrome P450 enzymes into a number of active metabolites
- Administration of CYP3A4 inhibitors may increase loperamide concentrations
- Loperamide side effects include constipation, dizziness, drowsiness, stomach pain, skin rash and itching
- Is not a DEA controlled medication
Diazepam
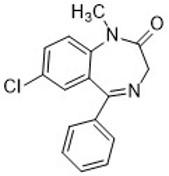
- Is a benzodiazepine used to treat anxiety associated with opioid withdrawal
- By binding to an allosteric site on the GABA A receptor, diazepam increases the effect of the neurotransmitter GABA
- Reduces anxiety and insomnia associated with opioid withdrawal
- Side effects include drowsiness and incoordination
- Excellent bioavailability
- Can lead to tolerance and dependence
- Must be tapered because abrupt stopping can be dangerous
Hydroxyzine
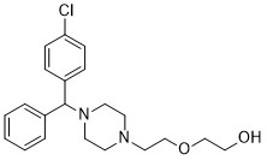
- Used to treat opioid withdrawal
- Is a histamine H1 receptor antagonist
- Reduces anxiety
- Causes sedation
- Produces antiemetic effects
- Side effects include drowsiness, incoordination, and dizziness
- Excellent bioavailability
- Metabolized by hepatic cytochrome P450 enzymes
Opioid Use Disorder
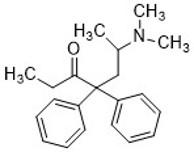
Buprenorphine
- Used for treating opioid abuse disorder, where it is combined with naloxone to prevent diversion of buprenorphine
- Is a partial agonist at the mu opioid receptor
- Administered sublingually because of poor bioavailability
- Metabolized by hepatic cytochrome P450 enzymes into a number of active metabolites
- Cleared by hepatic elimination
- Side effects include nausea, vomiting, drowsiness
- May cause respiratory depression but less than a full opioid agonist
- May lead to tolerance and dependence
- Discontinuation may lead to withdrawal symptoms
- DEA Schedule III drug
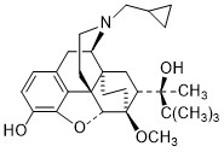
Methadone
- Used for opioid maintenance therapy in patients with opioid abuse disorder
- Has been used to treat pain
- Good oral bioavailability and is usually given orally
- Metabolized very slowly by hepatic cytochrome P450 enzymes leading to a long half-life
- Excreted by renal elimination
- May cause respiratory depression and death
- May lead to tolerance and dependence
- May cause constipation and urinary retention
- Prolongs the cardiac QT interval, which may lead to abnormal heart rhythm
- Discontinuation may lead to withdrawal symptoms
- DEA Schedule II drug
Antagonists
Naloxone
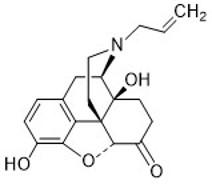
- Used to treat opioid overdose
- Is a nonselective antagonist at all opioid receptors
- Will induce withdrawal symptoms in an opioid-dependent person
- Is combined with buprenorphine to decrease the risk of opioid misuse when buprenorphine is being used to treat opioid use disorder
- Poor bioavailability. Administered by injection or nasal spray
- In opioid-naïve people, naloxone has very few side effects
- Metabolized by hepatic cytochrome P450 enzymes and glucuronidation
- Excreted by renal elimination
- Short half-life relative to other opioids
- Not DEA scheduled
Naltrexone
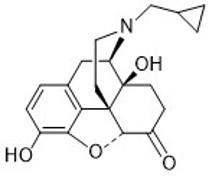
- Used to treat opioid use disorder and alcoholism
- Is a nonselective antagonist at all opioid receptors
- Will induce withdrawal symptoms in an opioid-dependent person
- Good bioavailability. Administered orally or as an injectable depot preparation
- The most common side effects are diarrhea and intestinal cramping
- Metabolized by hepatic cytochrome P450 enzymes and glucuronidation
- Excreted by renal elimination
- Not a DEA scheduled drug
Cannabidiol (CBD) in Pain Management
Cannabis
- Cannabidiol (CBD)
- Pharmaceutical grade CBD (Epidiolex®) Schedule 5 controlled substance (CS)
- Dietary supplement CBD (not regulated by FDA)
- Tetrahydrocannabinol (THC)
What is CBD?
- Non-psychoactive component of marijuana (vs. THC)
- Widely available dietary supplement with minimal regulations
- Consumer market $600 million - $2 billion in 2018
- Pharmaceutical grade CBD (Epidiolex® oral solution)
- FDA approved for Dravet syndrome and Lennox-Gastaut Syndrome in July 2018
For more: Cowen’s collective view of CBC. Report by Cowen Research, Feb. 29, 2019.
Highlights of prescribing information on Epidiolex®
CBD Product Labeling Accuracy
- 84 CBD extracts purchased online between 9/12/2016-10/15/2016
- Sent to lab for high performance liquid chromatography analysis
- THC detected in 18 samples (21.43%)
- Median labeled CBD concentration 15 mg/ml (Range: 1.33-800 mg/ml)
- Median observed CBD concentration 9.45 mg/ml (Range: 0.1-655.27 mg/ml)
- 36 underlabeled products (42.85%)
- 22 overlabeled products (26.19%)
- 26 accurately labeled products (30.95%)
- Considerable variability in labeled and observed CBD concentrations in supplements purchased online
Reference
Bonn-Miller MO, et al. Labeling accuracy of Cannabidiol extracts sold online. JAMA. 2017 Nov 7;318(17):1708-1709. doi: 10.1001/jama.2017.11909.
Studied CBD Indications
- Refractory epilepsy
- Dravet Syndrome
- Lennox-Gastaut Syndrome
- Schizophrenia
- Crohn’s disease
- Type 2 diabetes
- Parkinson’s disease
- Bipolar disease
- Social anxiety disorder
- Ocular HTN
- Chronic pain
- Huntington’s disease
- Fatty liver disease
- Graft versus host disease
- Dystonic movement disorders
Reference
Millar SA, et al. A systematic review of cannabidiol dosing in clinical populations. Br J Clin Pharmacol. 2019 Jun 20. doi: 10.1111/bcp.14038. [Epub ahead of print]
CBD Pharmacology
- Small affinity for CB1 and CB2 receptors
- May inhibit THC binding at CB1 receptor
- Transient receptor potential channels (TRPV1, TRPV2, TRPM8, TRPA1)
- G-protein coupled receptors (GPR55, GPR18)
References
- Samanta D. Cannabidiol: A review of clinical efficacy and safety in epilepsy. Pediatr Neurol 2019; 96:24-29. doi: 10.1016/j.pediatrneurol.2019.03.014 Epub 2019
- Lucas CJ, et al. The pharmacokinetics and the pharmacodynamics of cannabinoids. Br J Clin Pharmacol 2018; 84(11):2477-2482. doi: 10.111/bcp.13710 Epub 2018
CBD Pharmacokinetics
- Lipophilic with poor oral bioavailability
- Rapidly distributes into highly perfused organs
- Hepatically metabolized via hydroxylation and glucuronidation
- Many potential drug-drug interactions via CYP3A4, CYP2B6, CYP2E1, CYP2C8, CYP1A2, CYP2D6, CYP2C9, CYP2C19, UGT1A9, UGT2B7
- Highly protein bound
- Half-life (oral): 2-5 days
- Accumulation with chronic use in adipose tissue
- Fecal elimination
References
- Lucas CJ, et al. The pharmacokinetics and the pharmacodynamics of cannabinoids. Br J Clin Pharmacol 2018; 84(11):2477-2482. doi: 10.111/bcp.13710 Epub 2018
- Samanta D. Cannabidiol: A review of clinical efficacy and safety in epilepsy. Pediatr Neurol 2019; 96:24-29. doi: 10.1016/j.pediatrneurol.2019.03.014 Epub 2019
- Brown JD, Winterstein AG. Potential adverse drug events and drug-drug interactions with medical and consumer cannabidiol (CBD) use. J Clin Med 8(7). Pii: E989. doi: 3390/jcm8070989.
CBD Adverse Effects
- Most common adverse effects start within the first 2 weeks of treatment
- Sedation/fatigue
- Decreased appetite
- Nausea/vomiting/diarrhea
- Weight loss
- Insomnia
- Increased risk of infection
- Elevated liver function tests
- Usually within the first 2 months of treatment; reversible upon stopping CBD
References
- Highlights of prescribing information on Epidiolex®
- Millar SA, et al. A systematic review of cannabidiol dosing in clinical populations. Br J Clin Pharmacol. 2019 Jun 20. doi: 10.1111/bcp.14038. [Epub ahead of print]
- Samanta D. Cannabidiol: A review of clinical efficacy and safety in epilepsy. Pediatr Neurol 2019; 96:24-29. doi:10.1016/j.pediatrneurol.2019.03.014 Epub 2019
CBD for Pain Management
- Lack of clinical trial data in pain management
- 8 clinical trials currently listed on www.Clinicaltrials.gov (9/7/2019)1-2
- “N of 1” study methodology with 34 adult, chronic pain patients
- 2 weeks baseline
- 2 weeks run-in (open label THC:CBD)
- 4 X 1 week crossover, double-blind randomized periods X 2
- 4 treatment arms with pharmaceutical grade cannabis based medicinal extracts (CBME)
- THC:CBD 2.5mg/2.5mg SL spray
- Placebo SL spray
- THC 2.5mg SL spray
- CBD 2.5mg SL spray
References
- NIH US National Library of Medicine. Cannabidiol/Pain Studies. Clinicaltrials.gov
- Notcutt W, et al. Initial experiences with medicinal extracts of cannabis for chronic pain: Results from 34 ‘N of 1” studies. Anaesthesia 2004; 59(5):440-452.
Summary of Notcutt et al. (2004)
- Primary aim: To identify therapeutic windows of 3 active CBMEs (THC:CBD, THC, CBD)
- Patients could select up to 5 symptoms including pain to evaluate 3 times/day on visual analogue scale
- Patients could use 1-8 sprays/dose, up to 6 times/day
- Results
- 24 patients completed study
- Variety of pain etiologies
- No improvement in symptoms (figure 5) or duration of sleep (figure 8)
- Improved sleep quality (figure 7)
- No patients preferred CBD monotherapy, THC:CBD preferred overall
- Averaged 9 sprays/day (figure 9)
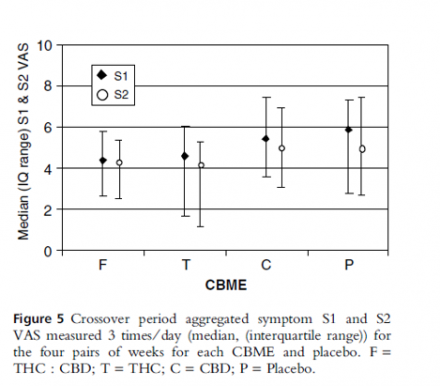
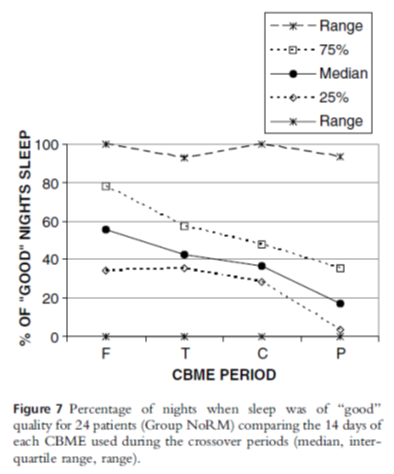
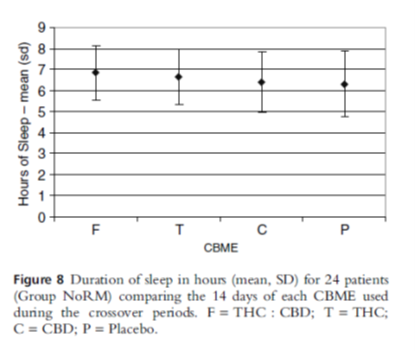
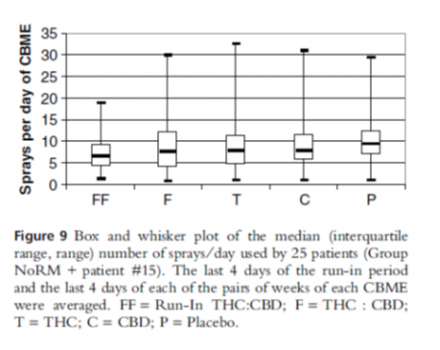
Reference
Notcutt W, et al. Initial experiences with medicinal extracts of cannabis for chronic pain: Results from 34 ‘N of 1” studies. Anaesthesia 2004; 59(5):440-452.
CBD Dosing
- May be a future role for CBD in pain management but current evidence does not support its use; high quality clinical trials needed
- Wide range of CBD dosing in the clinical literature for various indications
- <1-50 mg/kg/day
- Extrapolation of dosing from other disease states indicates better therapeutic outcomes with higher doses
- Average CBD dose of 22.5mg/day in study by Notcutt et al. 2004 may be subtherapeutic and potentially contributed to limited clinical benefit
References
- Millar SA, et al. A systematic review of cannabidiol dosing in clinical populations. Br J Clin Pharmacol. 2019 Jun 20. doi: 10.1111/bcp.14038. [Epub ahead of print]
- Notcutt W, et al. Initial experiences with medicinal extracts of cannabis for chronic pain: Results from 34 ‘N of 1” studies. Anaesthesia 2004; 59(5):440-452.
Pathophysiology and Non-Opioid Management of OA
Osteoarthritis (OA)
- Inflammatory
- Degenerative joint disease
- The most common form of arthritis
- Cartilage in the joints wears down
- Most commonly affects joints in
- Hands
- Hips
- Knees
- Spine
OA: Risk Factors
- Age
- Risk increases with age
- Sex
- Women are more likely to develop OA
- Obesity
- Added stress on weight-bearing joints
- Fat tissue produces proteins that may cause inflammation
- Joint injuries
- Ex. from playing sports or from an accident
- Certain occupations
- Especially if it requires repetitive stress on a particular joint
- Genetics
- Bone deformities
- Other diseases
- Diabetes, gout, rheumatoid arthritis (RA)
Reference
Mayo Clinic. Patient care and health information: Diseases and conditions. Osteoarthritis
OA-Pathophysiology
- Under normal conditions the joint matrix is subjected to a dynamic remodeling process
- Low levels of degradative and synthetic enzyme activities are balanced
- The volume of cartilage is maintained
- In OA cartilage, however, matrix degrading enzymes are overexpressed, shifting this balance in favor of net degradation
- Various cytokines (IL-1, TNF-α) stimulate the production of metalloproteinases
- Metalloproteinases degrade cartilage and also inhibit the production of collagen and matrix
Joint Physiology - OA
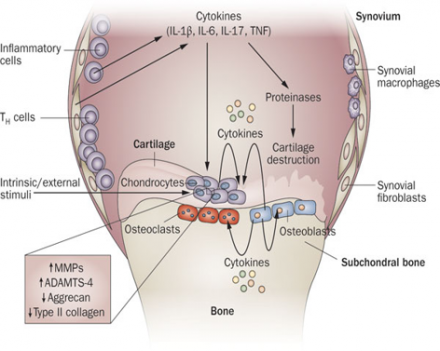
The levels of proinflammatory cytokines, including IL-1β, TNF and IL-6, are elevated in OA. These cytokines contribute to the pathogenesis of OA through several mechanisms, including downregulation of anabolic events and upregulation of catabolic and inflammatory responses, effects that result in structural damage to the OA joint. Abbreviations: ADAMTS, a disintegrin-like and metalloproteinase with thrombospondin type 1 motifs; IL, interleukin; MMP, matrix metalloproteinase; OA, osteoarthritis; TNF, tumor necrosis factor.
Reference
Kapoor M, Matel-Pelletier J, Lajeunesse D, Pelletier J-P, Fahmi H. Cytokines in the pathophysiology of osteoarthritis. Nature Reviews 2011; 7:32-42.
Treatment of OA Pain: Non-Opioid Agents
Aminophenol (or APAP = acetyl-para-aminophenol)
- Acetaminophen (Tylenol®)
NOTE: This is NOT an NSAID, but does have analgesic and antipyretic effects similar to those of aspirin.
- Indications: pain, fever, OA (*not a labeled indication)
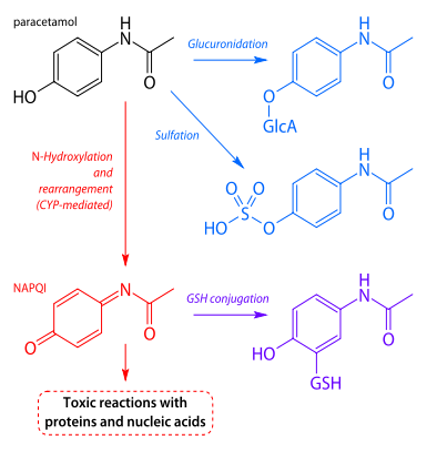
- Metabolism:
- Mostly glucoronidation and sulfate conjugation (80-90%)
- Oxidative metabolism via 2E1 & 1A2 toxic metabolite (NAPQI)
- Inactivated by glutathione
- With large doses, main route is saturated
- FDA not more than 325 mg of acetaminophen per dose in combination products
*Hochberg MC, et al. American College of Rheumatology 2012 recommendations for the use of non-pharmacologic therapies in osteoarthritis of the hand, hip, and knee. Arthritis Care & Research 64(4):465-474.
Agency for Healthcare Research and Quality (AHRQ): Guidelines and measures.
NSAIDs
Boxed Warning for NSAIDs
- Increased risk of CV events
- MI, stroke, hypertension
- May compromise renal function
- Risk of GI irritation
Class Side Effects/Warnings
- Avoid alcohol, compounding effect on GI problems
- Take with food, milk, antacid to decrease GI effects
- Pregnancy D: 3rd trimester
- May inhibit uterine contractions
- Premature closure of ductus arteriosus
- January 2015 FDA special alert
- “The U.S. Food and Drug Administration (FDA) is aware of and understands the concerns arising from recent reports questioning the safety of prescription and over-the-counter (OTC) pain medicines when used during pregnancy. As a result, we evaluated research studies published in the medical literature and determined they are too limited to make any recommendations based on these studies at this time. Because of this uncertainty, the use of pain medicines during pregnancy should be carefully considered. We urge pregnant women to always discuss all medicines with their healthcare professionals before using them."
Pregnancy
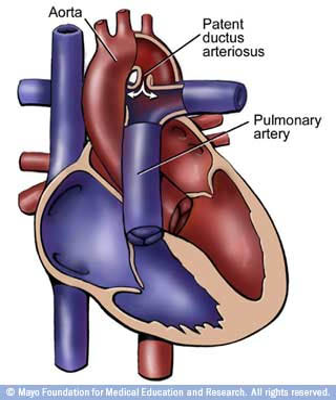
- Prostaglandins (particularly E and F series) stimulate uterine contractions
- Abortifacient
- Both cyclooxygenase enzymes produce this effect
- Ductus Arteriosus
- Prostaglandins are responsible for maintaining the ductus arteriosus
- Allows blood from the right ventricle to bypass the fetus’ fluid filled lungs
Major Adverse Effects of NSAIDs
| Adverse Effect | Non-Selective COX Inhibitors | COX-2 Selective Inhibitors |
|---|---|---|
| Gastric ulceration | Yes | Yes* |
| Inhibit platelet function | Yes | No |
| Inhibit labor induction | Yes | Yes |
| Impaired renal function | Yes | Yes |
| Hypersensitivity reaction | Yes |
? |
*The GI toxicity of COX-2 selective inhibitors may be less than that of
non-selective COX inhibitors, but there is still some incidence of toxicity
Reference
Golan DE, Armstrong EJ, Armstrong AW. Principles of Pharmacology. The Pathophysiologic Basis of Drug Therapy. Wolters Kluwer, 4th ed. Diagram: LWW, Principles of Pharmacology, /2e (Golan)
Things to keep in mind with NSAIDs
- Contraindications:
- Pain following CABG
- Heart failure
- Aspirin triad (eg, bronchial asthma, aspirin intolerance, rhinitis
- Precautions:
- HTN
- Hepatic impairment
- History of GI disease (bleeding, ulcers)
- Renal impairment
- Drug Interactions:
- Antihypertensives (ACEI, ARBs, hydralazine, beta blockers, etc.): decreased effectiveness
- Lithium: Increased lithium concentration
- SSRIs: enhance anti-platelet effect of NSAID
- Bisphosphanates (Fosamax): Increased risk of GI irritation
- Ethanol: enhanced GI irritation
Propionic Acid Derivatives
- Ibuprofen (Advil®, Motrin®)
- Naproxen (Aleve®, Naprosyn®, Anaprox®)
- Ketoprofen (Orudis®, Oruvail®)
- Flurbiprofen (Ocufen®, Ansaid®)
- Oxaprozin (Daypro®)
- Fenoprofen (Nalfon®)
Ibuprofen (Advil®, Motrin®)
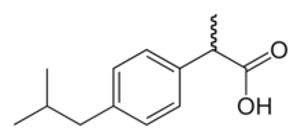
- Indications:
- Mild to moderate pain
- Fever
- Headache
- JRA/JIA
- OA; RA
- Metabolism: oxidation
- Inhibitor of CYP2C9
- Onset of action: 30-60 min
- t½: 2-4 hr.
- Formulations:
- Oral (tabs, caps, susp, drops)
- IV (Caldolor®)
Naproxen (Aleve, Anaprox, Naprosyn)
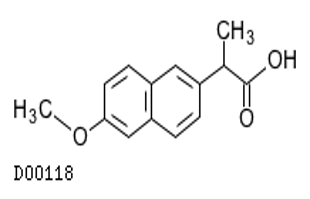
- Indications:
- Mild to moderate pain
- Fever
- Headache
- Ankylosing spondylitis
- JRA/JIA
- OA; RA
- Metabolism:
- Inhibits CYP1A2 and 2C9
- t½: 12-17 hrs.
- Onset of action:
- 30min for sodium
- 1hr for base
- Formulation: oral (tabs, caps, susp)
Oxaprozin (Daypro®)
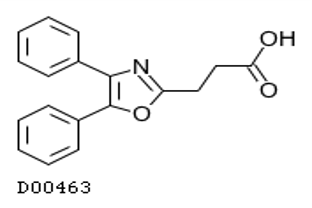
- Indications:
- JRA/JIA
- OA; RA
- Metabolism: oxidation and glucuronidation
- t½: 40-50 hrs.
- Once daily dosing
- Onset of action: 2-4 hrs.
- Formulations: oral
Indole Acetic Acid Derivatives
- Indomethacin (Indocin®)
- Sulindac (Clinoril®)
- Etodolac (Lodine®)
Indomethacin (Indocin®)
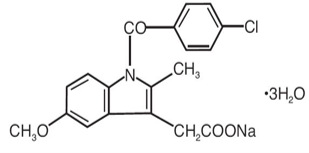
- Indications:
- Moderate to severe pain
- Ankylosing spondylitis
- Gouty arthritis
- OA; RA
- Patent ductus arteriosus
- Metabolism: CYP2C9, 2C19 (minor)
- Strong CYP2C9 inhibitor
- Significant enterohepatic recirculation
- t½: 4 hrs.
- Prolonged in neonates
- Onset of action: 30 min.
- Formulations:
- Oral (caps, susp)
- IV
- Suppository
Etodolac (Lodine®)
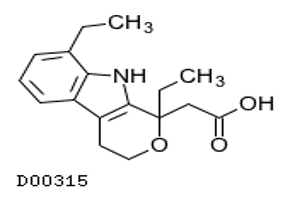
- Indications:
- Mild to moderate pain
- JRA/JIA
- OA; RA
- Metabolism: hydroxylation
- Onset of action:
- IR: 30 min.
- ER: 2-4 hrs.
- t½:
- IR: 6-7 hrs.
- ER: 8 hrs.
- 6-16yrs: 12 hrs.
- Formulations: oral
Phenyl Acetic Acid Derivative
- Diclofenac (Voltaren®, Cataflam®, Zipsor®)
- Ketorolac (Toradol®, Acuvail®, Acular®)
Diclofenac (Voltaren®, Cataflam®)
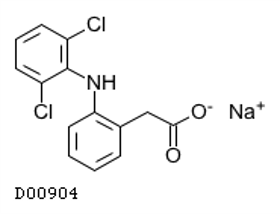
- Indications:
- Mild to moderate pain
- Ankylosing spondylitis
- Migraine
- OA; RA
- Postoperative ocular inflammation; ocular pain
- Metabolism: CYP2C9, 3A4, 1A2, 2D6
- Inhibits 1A2, 3A4 (weak)
- Extensive first pass metabolism
- Onset of action: 1-2 hrs.
- t½: 2 hrs.
- Patch: 12 hrs.
- Formulations:
- oral, gel, patch, opthalmic
Ketone Derivatives
- Nabumetone (Relafen®)
Nabumetone (Relafen®)
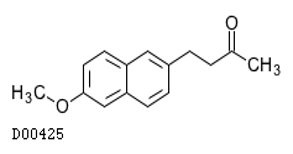
Oxicam Derivative
- Meloxicam (Mobic®)
Meloxicam (Mobic®)
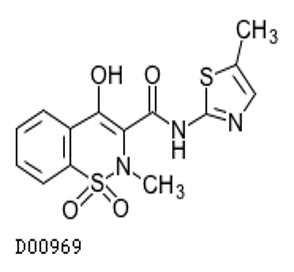
- Indications:
- JRA/JIA
- OA; RA
- Metabolism: CYP 2C9; 3A4 (minor)
- Inhibits CYP2C9
- Onset of action: 5-10 hrs.
- t½: 15-20 hrs.
- Can be dosed once daily
- Formulations: oral (tabs, susp)
COX2
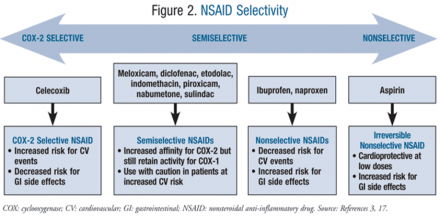
Reference
Perry LA, Mosler C, Atkins A, Minehart M. Cardiovascular risk associated with NSAIDs and COX-2 inhibitors. U.S. Pharm 2014; 39(3):35-38.
COX-2 and Cardiovascular Risk
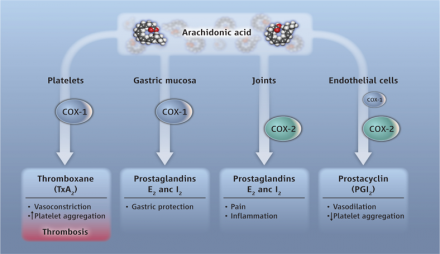
For more: http://www.sciencemag.org/content/336/6087/1386/F1.expansion.html
COX-2 Inhibitor
- Celecoxib (Celebrex®)
- Only “selective” COX-2 inhibitor
Celecoxib (Celebrex®)
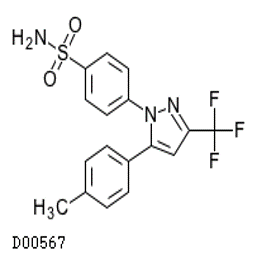
- Indications:
- Acute pain
- Ankylosing spondylitis
- Primary dysmenorrhea
- JIA
- OA/RA
- Metabolism: CYP2C9
- Inhibits CYP-2CA, 2D6
- Onset of action: 3 hrs.
- t½: 11 hr.
- Can be dosed once daily depending on indication
- Excretion:
- Urine and feces
- Formulation: oral
- Considerations:
- Caution in patients with sulfonamide hypersensitivity
- Caution in African Americans due to possible increase in systemic exposure (40% increase)
- Increase in systemic exposure (40-50%) in elderly
- Especially women (due to bodyweight)
- Juvenile RA: cardiovascular risk is unknown
Adjuncts
Steroid Injection
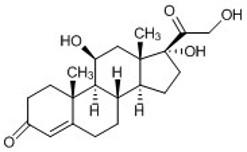
- Intra-articular steroid injections are given to reduce inflammation and associated pain
- Cortisol is produced naturally by the adrenal glands
- Suppresses the immune system, thereby reducing inflammation
- The most common side effect is pain and swelling in the joint where the injection was given
- Because the hydrocortisone injection is localized, there is little risk for severe side effects
Chondroitin
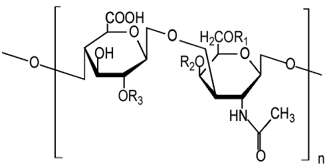
- A supplement for osteoarthritis that is often used with glucosamine
- Occurs naturally in connective tissue
- Important structural component of cartilage
- Composed of a chain of alternating sugars, N-acetyl-galactosamine and glucuronic acid
- Acts as an anti-inflammatory
- Stimulates the synthesis of proteoglycans and hyaluronic acid
- No major side effects
- Good bioavailability
Glucosamine
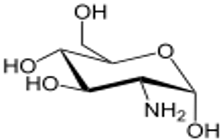
- An amino acid that is a precursor for the synthesis of glycosylated proteins
- A marketed supplement to support the structure and function of joints
- Insufficient evidence that this treatment is helpful with osteoarthritis
- Should not be used in people taking a blood thinner or in people receiving chemotherapy
- Adverse effects may include vomiting, diarrhea or constipation, and headache

