Migraine Treatment
Jean M. Bidlack, PhD
David J. Hutchinson, PharmD
Jennifer L. Mathews, PhD
Learning Objectives
- Describe goals of therapy for migraine treatment
- List common non-pharmacologic approaches to the treatment of migraine
- Discuss the drug treatment options and dosage forms available for migraine treatment
- Describe the mechanism of action of each class of medication
- Discuss medications used for migraine prophylaxis and explain their mechanisms of action
Goals of Any Therapy
- Reduce the frequency and severity of headaches
- Reduce headache-related disability
- Reduce reliance on pharmacotherapy that may be poorly tolerated or unwanted
- Promote the patient’s personal control of migraine
- Reduce headache-related distress and physical symptoms
Non-Pharmacologic Options: Therapies and Lifestyle Modifications
| Therapies | Lifestyle Modifications |
|---|---|
|
|
Non-Pharmacologic Options: Indications and General Principles
| Indications | General Principles |
|---|---|
|
|
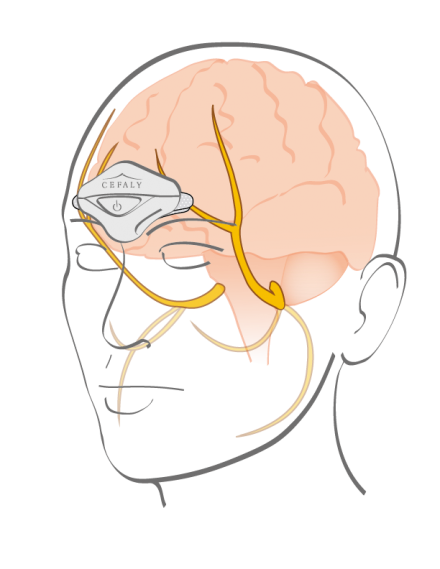
Cefaly®
- TENS
- Transcutaneous electrical nerve stimulation
- Approved March 2014
- Mechanism of Action: The device applies an electric current to the skin and underlying body tissues to stimulate branches of the trigeminal nerve, which has been associated with migraine headaches
Image
Pharmacologic Therapy
Image
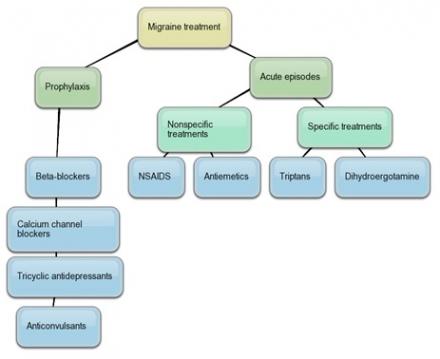
Goals of Pharmacologic Therapy
| Acute (Abortive) | Preventive (Prophylactic) |
|---|---|
|
|
- Allow patient to self-treat
- Reduce disability, improve quality of life
- Minimize adverse effects of therapy
Question: All migraine patients should be prescribed both acute and prophylactic treatment
Incorrect
Incorrect
Correct
Correct
Acute Therapy Only
- Migraines are infrequent
- Short in duration
- Responsive to acute medication therapy used after onset of attack
- Not significantly
Acute + Preventive
- Preventive therapy:
- Frequent disabling headaches – ≥2 times per month
- Frequent abortive therapy use – ≥2 times per week
- Recurring headaches that significantly disrupt daily life
- May be used in patients who do not respond well to abortive medication(s)
- Intolerance, lack of efficacy, comorbidities/contraindications
Acute episodes: Proactive vs. Reactive Stepped Care/Stratified Care
| Stepped Care | Stratified Care |
|---|---|
|
|
Acute Non-specific: Analgesics and NSAIDs
- Non-specific therapy
- Many are available OTC
- Relatively inexpensive
- Combination products
- Ex: Excedrin Migraine
acetaminophen 250mg + Aspirin 250mg + Caffeine 65mg
- Risk of medication overuse headache
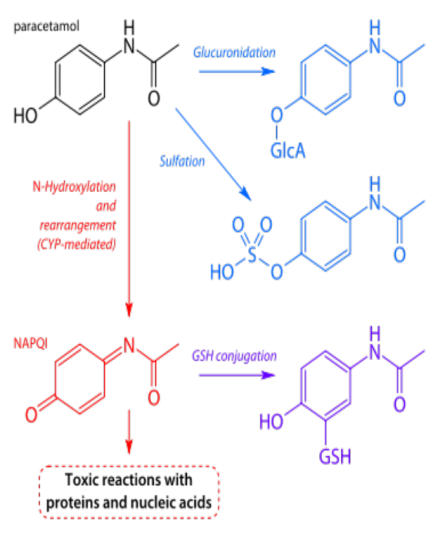
Acetaminophen† (Tylenol®)
- 500mg-1g ever 6 hours
- MDD: 4g/day
- Metabolism: hepatic
- 85-90% glucuronidation/conjugation
- 10-15% CYP2E1 and CYP1A2
- Onset: 30 minutes
- Used alone or in combination
Image
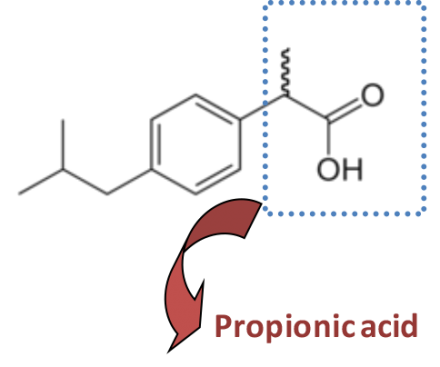
Ibuprofen (Advil®, Motrin®)
- Metabolism: oxidation
- Inhibitor of CYP2C9
- Onset of action: 30-60 min
- t½: 2-4 hr
- 800mg every 6 hours
- MDD: 2.4g/day
Image
Acute: Migraine-Specific Drugs
- Ergot Alkaloids/Ergotamines
- Triptans
Ergot alkaloids (Ergotamines)
- Derivatives of Claviceps purpurea (fungus)
- First migraine-specific drugs
- Germany 1893, US and UK 1894
- Historically, used both acutely and for prophylaxis
- Not used frequently
- Dependence
- Toxicity
Image

Ergotamines
- Indications: Migraine with or without aura
- Mechanism of Action: Complex!
- Alpha-adrenergic: partial agonists, antagonize Norepinephrine/5-HT (serotonin)
- 5-HT receptors: partial agonist
- Dopamine receptors: partial agonist
- Net result = cerebral and peripheral vasoconstriction
Ergotamine
- Low oral bioavailability + >90% first pass effect
- Formulations: SL tablet, suppository (with caffeine)
- Avoid in Pregnancy
- Vasoconstriction of uterine vessels, decreased placental blood flow
- Onset = 0.5-3 hours
- T½ = 2 hours
- Excretion
- Primarily fecal (90%)
- Metabolism: CYP3A4
- Adverse Drug Reactions: N/V, dizziness, bradycardia
- Dose: 2mg every 30 minutes
- Max 6mg/24 hours, or 10mg/week
- Caffeine enhances absorption of ergotamines; there are also combination oral formulations
Dihydroergotamine (Migranal®)
- Mechanism of Action: Similar to parent compound
- Increased alpha-adrenergic blockade
- Improves tolerance
- Pregnancy - contraindicated
- Adverse Drug Reactions: N/V, dizziness, rhinitis (nasal spray)
- Low bioavailability, >90% First Pass Effect (FPE)
- Onset = 15-30 min (IM)
- T½ = 9-10 hr
- Metabolism: CYP3A4
- Excretion: primarily fecal
- Formulations: IV, IM, SubQ, nasal spray
Question: Which of the following patients should NOT take dihydroergotamine? Select all that apply.
Correct
Correct
Correct
Correct
Correct
Correct
Incorrect
Incorrect
Triptans
- Almotriptan (Axert®)
- Eletriptan (Relpax®)
- Rizatriptan (Maxalt®)
- Sumatriptan (Imitrex®)
- Zolmitriptan (Zomig®)
- Migraine-specific drugs
- 5-HT1B/1D agonists
- Inhibit calcitonin gene-related peptide (CGRP) in trigeminal sensory neurons
- Abortive therapy for acute migraine episode
- Most effective when taken early in the course of the attack (mild pain)
- NOT indicated for migraine prophylaxis
- Guideline exceptions: Frovatriptan, Naratriptan, Zolmitriptan
- Short-term prophylaxis for menstrually-related migraine (MRM)
- Level A or B
Triptans: Contraindications
- Cardiovascular: CAD/IHD/MI, arteriosclerosis, angina, PVD, HTN
- Cerebrovascular: Cerebrovascular disease, stroke, basilar or hemiplegic migraine
- Severe hepatic disease
- Concurrent Monoamine Oxidase Inhibitor (MAO-I) therapy
Triptans: Precautions
- Pregnancy Category C
- 24hr washout
- Triptan and ergot
- Triptan and other 5-HT agonists – ex. Amphetamines
- Triptan and another triptan
- 2 week washout between triptan and monoamine oxidase inhibitors (MAO-I)
- Dose adjust in renal/hepatic impairment
- Common adverse drug reactions of triptans:
- Tight feeling in throat or chest
- Flushing; parasthesias
- Sleepiness
Sumatriptan (Imitrex®)
Image
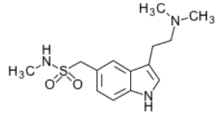
- First triptan available
- Indications: migraine +/- aura
- Onset = 30 minutes (oral)
- T½ = 2-3 hr
- Metabolism: hepatic, primarily monoamine oxidase A (MAO-A)
- Excretion: primarily urine
- Formulations: SQ, PO, intranasal, transdermal
- Treximet®: combination with naproxen
- = sumatriptan 85mg + naproxen 500mg
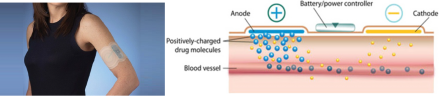
Sumatriptan: Novel Dosage Form
- Zecuity®
- Transdermal iontophoretic patch
- Why is a transdermal dosage form useful?
Image
Rizatriptan (Maxalt®)
Image
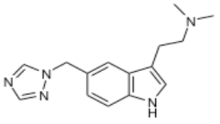
- Indications: migraine +/- aura
- Onset = 2 hr
- T½ = 2-3 hr
- Metabolism: MAO-A
- Excretion: primarily urine
- Formulations: PO tablet, Orally Disintigrating Tablets (ODT)
Rizatriptan: Clinical Pearls
- Maxalt-MLT contains phenylalanine
- Important to know if patient has PKU
- Drug interaction with propranolol
- Concurrent administration of propranolol 240 mg/day and a single dose of rizatriptan 10 mg resulted in a 70% increase in the mean rizatriptan AUC.
- Dose adjust rizatriptan
- Changes max to 5mg/dose, Major Depressive Disorder (MDD) 15mg/24h
Eletriptan (Relpax®)
Image
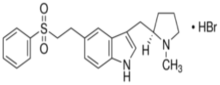
- Indications: migraine +/- aura
- Onset = 1.5-2 hours
- T½ = 4 hrs (metabolite 13hrs)
- Metabolism: CYP3A4 to active metabolite
- Excretion
- Formulations: PO tablet
Triptans: Major Depressive Disorder
- Dosing for triptans
- All labeled: May repeat in 2 hr* if the migraine headache has not resolved
- Controlled trials have not established the effectiveness of a second dose if the initial dose was ineffective
- All have MDD
- Almotriptan (Axert®):
- MDD 25 mg
- Eletriptan (Relpax®):
- MDD 80 mg
- Frovatriptan (Frova®):
- MDD 7.5 mg
- Naratriptan (Amerge®):
- MDD 5 mg (*repeat in 4 hrs)
- Rizatriptan (Maxalt®):
- MDD 30 mg (*propranolol)
- Sumatriptan (Imitrex®):
- MDD 200 mg
- Zolmitriptan (Zomig®):
- MDD 10 mg
(Clinical Pharmacology and LexiComp)
CGRP Antagonists
- Several have been tested
- Many with limiting side effects, particularly liver toxicity
- Rationale makes sense when considering the role of CGRP in migraine etiology
- Theoretically better than serotonin agonists, which can be problematic particularly with cardiovascular disease
- For more information:
Other Pharmacologic Options
- Barbiturates: butalbital†
- Opioids: butorphanol (others off-label)
- Anticonvulsants†
- Midrin® (acetaminophen/dichloralphenazone/isometheptene)
- Midrin: functionally discontinued – no longer being produced
- Can be compounded
- Complementary and Alternative Medicine (CAM)†
Feverfew
Image

- Tanacetum parthenium
- Chrysanthemum species
- Mechanism of Action: unknown
- Conflicting data regarding efficacy
- Several pain/inflammatory pathways have been studied without definitive results
- Avoid in pregnancy: risk of uterine contractions
- Avoid in breast-feeding: limited data
Antiemetics
- Adjunct therapy, if N/V is a component of migraine
- 5-HT3 antagonists
- ondansetron, alosetron, dolasetron, granisetron, palonosetron
- Phenothiazines
- promethazine, prochlorperazine, perphenazine
- Antihistamines
- cyclizine, hydroxyzine, meclizine
- Dopamine antagonist
- metoclopramide
- Dexamethasone
- Mechanism of action as antiemetic unknown
Assessing Migraine Therapy
Image

Migraine Prophylaxis
Indications:
- More than 2 headaches per month
- Poorly managed with acute treatment options
- Contraindication(s)
- Intolerance
- Medication overuse
- Treatment failure
- Consider cost of acute and preventive therapies
- Uncommon migraine conditions
- Basilar, hemiplegic, migraine with prolonged aura
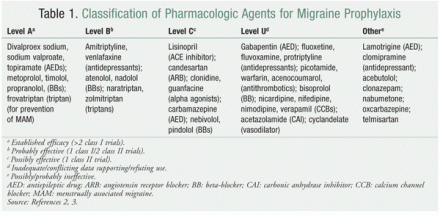
Prophylactic agents
- Levels A-B
- Antiepileptics
- Beta-blockers
- Others
- Calcium channel blockers
- ACE-Is/ARBs
- Anti-histamines
Image
Antiepileptics
- Valproic Acid (level A)
- Topiramate (level A)
- Valproate (hypotheses)
- Increases GABA transmission
- Blocks voltage-gated sodium channels
- Modulates release of Dopamine and 5-HT
- Other antiepileptics block voltage-gated sodium channels potentiate GABA receptor activity
Beta-blockers
- Propranolol (level A)
- Timolol (level A)
- Metoprolol†* (level A)
- Atenolol†* (level B)
- Timolol†* (level B)
- Mechanism of Action: thought to modulate the catecholamine responses produced in migraine patients
- Anxiolytic properties address the stress-related component of migraine (locus coeruleus)
*Off-label, but found in treatment guidelines
Onabotulinumtoxin A (BOTOX®)
- Botulinum toxin A
- Neurotoxin
- MOA (hypothesis):
- Interrupts neuromuscular signaling
- Inhibits release of excitatory amino acids, and inflammatory neuropeptides
- Qualifying for treatment:
- Frequency: patient must meet criteria for “chronic migraine,” defined as migraine headaches ≥15 days/month,
- PLUS
- Duration: headaches last ≥4 hours per day
- 18 or older
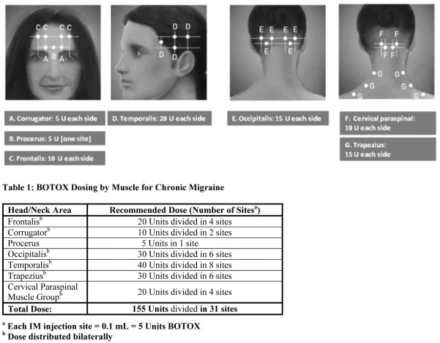
Onabotulinumtoxin A (BOTOX®)
- Tx: prophylactic treatment of chronic migraine
- Dose: IM 5 units/0.1mL per site
- Total dose: 155 units once every 12 weeks (bilaterally) into 31 total sites
- Problems swallowing, speaking, or breathing, due to weakening of associated muscles, can be severe and result in loss of life. Swallowing problems may last for several months.
- Spread of toxin effects. The effect of botulinum toxin may affect areas away from the injection site and cause serious symptoms including: loss of strength and all-over muscle weakness, double vision, blurred vision and drooping eyelids, hoarseness or change or loss of voice (dysphonia), trouble saying words clearly (dysarthria), loss of bladder control, trouble breathing, trouble swallowing.
Botox injection sites: migraine
Image
General Approaches to Pharmacological Treatment
- Consider tolerability of therapy for the patient
- Maintain realistic expectations (patient and provider)
- Give therapy an adequate trial – 2 months
- Can be hard for a patient who is in pain
- Non-pharmacologic interventions can be used concurrently to maximize results
- Make changes at that time if necessary

